 ET todayĬambridge, MA - JanuLeap Therapeutics, Inc. The marker is expressed in GI and pancreatic cancers.Leap to host conference call at 9:00 a.m. Other companies are also focused on Claudin18.2, including Turning Point Therapeutics, Elevation Oncology and AstraZeneca. In the conference call, Onsi said, “We believe we have found the perfect fit with Flame, identifying a second clinic-ready antibody program, expanding our GI focus, building on our biomarker expertise and development strategy of building personalized medicines to a targeted patient population.” 
It was clear that the Leap development team, with its expertise in developing DKN-01, was the ideal partner for FL-301, our preclinical assets, and the Flame shareholders.” In a press statement, Martin said, “Flame conducted an extensive strategic process. Richard is Head of Public Research at Samsara BioCapital. Leap picked up two more board members nominated by Flame, Patricia Martin and Christian Richard. The Leap team will handle all executive positions of the combined company. 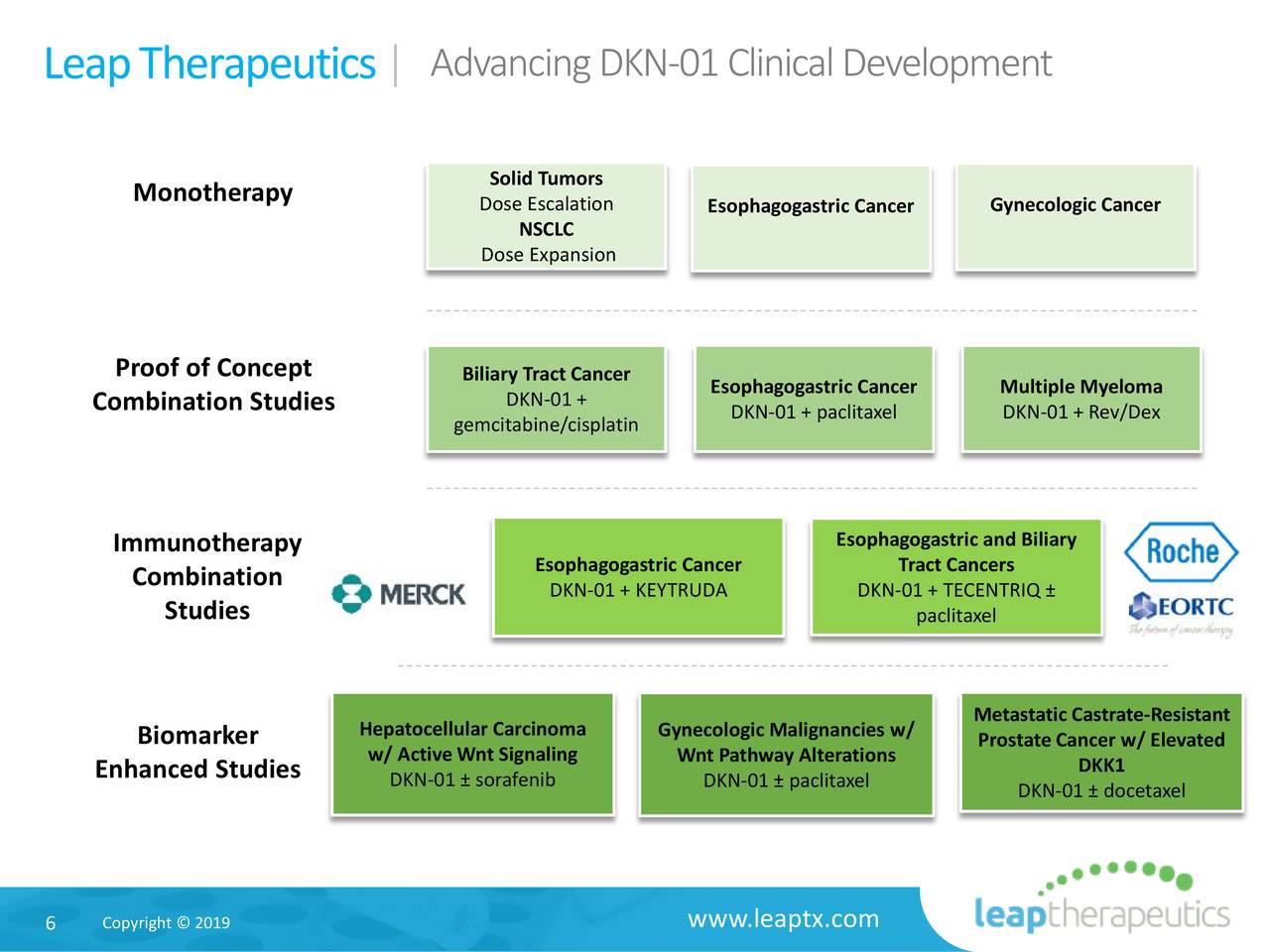
Macrocure became a wholly owned subsidiary and Leap went public. Leap itself went public in 2016 in a reverse merger with Macrocure. Under the transaction, Flame shareholders will hold approximately 58% of outstanding shares. The company is a private, venture-backed cancer company with support from Samsara BioCapital, Rock Springs Capital, Janus Henderson, Adage Capital Management, Surveyor Capital, Cormorant Asset Management and others. However, in the fall of 2022, abandoned the project after Novartis’ drug in the same class flunked several clinical studies. 
Leap holds exclusive rights for development, manufacturing, and commercialization of the compound for the rest of the world.ĭKN-01 is also being developed with Merck’s Keytruda and Genentech’s Tecentriq for gastroesophageal adenoma and advanced esophagogastric adenocarcinoma, respectively.įlame Biosciences originally focused on a monoclonal antibody IL-1ß inhibitor. Leap and BeiGene inked an exclusive option and license deal for DKN-01 in Asia (except Japan), Australia, and New Zealand. The drug has been granted Orphan Drug Designation by the FDA for gastric and gastroesophageal junction cancer as well as Fast Track Designation in combination with BeiGene’s anti-PD-1 checkpoint inhibitor tislelizumab for gastric and gastroesophageal junction adenocarcinoma tumors that highly express DKK1. Leap’s own lead program is DKN-01, a humanized monoclonal antibody targeting DKK1, which modulates the Wnt/Beta-catenin and PI3kinase/AKT signaling pathways. 
Leap issued shares of its common stock to Flame shareholders and agreed to pay the shareholders 80% of after-tax proceeds from any licensing deals of Flame’s early-stage compounds, FL-101 or FL-103. Leap also acquired Flame’s net cash, which Onsi said “was approximately $50 million as of year-end.” Together the combined company will have about $115 million in cash, sufficient to fund the expanded pipeline into 2025. In addition to FL-301, Leap acquired Flame’s FL-302, a preclinical anti-Claudin18.2/CD137 bispecific monoclonal antibody, and FL-501, a preclinical anti-GDF15 monoclonal antibody. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
